With 127 millions diabetic adults in China alone—almost a quarter of the global diabetic population—the country’s experience with the diabetes epidemic has become a health disaster.
To begin filling the need for tailored solutions, last year Chinese pharmaceutical giant Hunan China Sun paid $2 million for a 20% stake in Glucovation, a San Diego-based life science SME developing an innovative continuous glucose monitoring (CGM) system for Chinese diabetics.
Following the recent signing of a $12.5 million licensing and development agreement between the two companies, a China-based joint venture began pre-clinical human trials of Glucovation technology in February.
We caught up with Glucovation executive vice president Fernando Corona to discuss the company and its China focus.
MedicalExpo e-Magazine: Can you tell us a little about Glucovation and what the company is currently working on?
Fernando Corona: Glucovation was founded in May 2013 by three ex-Dexcom (another California-based CGM company) employees. We are currently developing a minimally invasive, low-cost CGM system that is factory calibrated (no daily finger sticks) for use in price sensitive markets such as China.
Our system uses a micro needle that is painless to insert, and which can be left in the abdomen or an arm for up to 10 days without discomfort. It samples interstitial fluid, in which the level of blood glucose correlates well to that of blood itself. The sensor we’re developing is non-enzymatic—its electrochemical properties achieve performance levels that have never been seen in the CGM space before.
Anyone affected with Type 1, Type 2 or pre-diabetes will be able to monitor their glycemic levels using the device.
Last October, it was revealed that Johnson & Johnson Animas OneTouch Ping insulin pump had a cybersecurity vulnerability. With the rising number of connected (mostly wireless) glucose meters and insulin pumps, cybersecurity is becoming a key concern in this industry.
In 2016, the Diabetes Technology Society published a protection standard called DTSec, which intends to decrease the threat level. It checks software integrity, data integrity and secure communication channel for connected diabetes devices.
Through DTSec, manufacturers can certify their products and guarantee a “good enough” level of cybersecurity. As explained in this article of the Journal of Diabetes Science and Technology, the standard is defining a maximum vulnerability rating that can’t be “expected to resist very prolonged attacks by very experienced hackers using very substantial resources.”
Three to five times a day, patients with diabetes have to prick their fingers to draw a drop of blood to determine if their blood glucose levels need to be adjusted.
The demands of the regimen can wear patients down, threatening their compliance with monitoring and, therefore, their health.
“In the diabetes community, it’s called ‘diabetic burnout.’ Patients just want to be free from the disease, and they can’t,” said Harry Gandhi, CEO and co-founder of Waterloo, Canada-based Medella Health.
Medella aims to prevent diabetic burnout with a non-invasive method to continuously monitor blood glucose levels with “smart lenses.”
Half-Dozen Companies At Work
Worldwide, about a half-dozen companies are developing smart lenses that contain a sensor. Those sensors measure the minute levels of blood glucose secreted in the eye and transmit the readings to a smartphone interface so patients can manage their levels with insulin, diet and exercise.
Probably the best-known smart lens developer is Alcon, a contact lens maker, which is a division of Novartis.
Gregory Herman, a chemical engineering professor at Oregon State University in Corvallis, Oregon, and his colleagues also have been developing sensors to measure glucose levels in the eyes using a transparent contact.
He said: “The idea behind these lenses is they are less intrusive than finger pricks. If people are already wearing contact lenses, for example, it just isn’t going to be that much of a hindrance to their lifestyle and they will not need to be pricking their finger after meals or before meals and make decisions.”
For Patient With Type 1 Diabetes
Herman, whose research is funded by JDRF (formerly the Juvenile Diabetes Research Foundation in New York City), said the lenses are ideal for patients with Type 1 diabetes. “For Type 1 diabetes, it’s best to do continuous glucose monitoring, which tracks the glucose levels throughout the day and night. This way the patient and doctor can get a lot more data as opposed to just pricking your finger several times a day,” Herman said. “This data will help make predictions based on someone’s history—for example, typical times for meals or physical activity.”
This data will help make predictions based on someone’s history—for example, typical times for meals or physical activity.
Google announced the lens in 2014 to great fanfare. Developers said they were testing a smart contact lens that’s built to measure glucose levels in tears using a tiny wireless chip and miniaturized glucose sensor that are embedded between two layers of soft contact lens material.
“We’re testing prototypes that can generate a reading once per second. We’re also investigating the potential for this to serve as an early warning for the wearer, so we’re exploring integrating tiny LED lights that could light up to indicate that glucose levels have crossed above or below certain thresholds,” they said.
The prototypes had a geeky Google look with visible monitors. Competitors are planning transparent lenses.
Verily Under Fire
The lenses came under fire in 2016 when a STAT investigation found that “the lens was regarded by many researchers—and even experts inside Verily—as technically infeasible because tears are not a reliable fluid for measuring blood sugar.”
Neither Verily nor its partner Novartis responded to MedicalExpo e-Magazine’s requests for interviews. An Alcon spokeswoman told STAT that the Verily lenses are in early clinical development and “for complex, cutting-edge technology, the projects are progressing steadily.”
Our big breakthrough was figuring out how to monitor glucose continually
Gandhi left the University of Waterloo, where the Medella project started, to focus on this technology and said his group’s sensors have been a success. “Our big breakthrough was figuring out how to monitor glucose continually, and in a way that it lasts for a very long time and simultaneously detects the minute levels of glucose with the basal tears,” he said.
Economic Issues
Herman said there is a fraction of glucose in the eyes compared with fluids under the skin. Still, he said the proper array of transparent sensors can accurately measure blood glucose levels accurately and reliably.
The science debate is crucial, but so are economic issues. Gandhi said the potential market in North America alone is $25 billion per year.
He said the smart lenses, which would be changed once a month, had to be priced about the same as current monitoring methods. That can be anywhere from $1,500 to $5,000 per year (in the U.S., with prices being significantly lower in Europe), depending on the brand and whether patients with Type 1 or Type 2 diabetes are being monitored (Type 1 patients require more testing).
Gandhi said he expects to know within a year when the lenses can be brought to market.
Herman’s group is focused on developing sensors rather than the entire device. “We should be able to make the contact lens just nominally more expensive. So I think at the end it would be a more cost-effective solution,” he said.
With diabetes now a global health issue of epidemic proportions, the need for better blood glucose monitoring techniques is more urgent than ever. Yet the typically invasive nature of monitoring, which involves finger pricks, tests strips and portable blood glucose meters, has caused problems.
Both type 1 and type 2 diabetics must monitor blood glucose regularly. This can involve 10 or more finger prick tests each day, although studies have shown that many diabetics avoid such tests completely, largely due to the pain and inconvenience.
In recent years the availability of continuous glucose monitoring (CGM) devices, which analyze interstitial fluid glucose levels in real time via subcutaneous sensors, has helped some diabetics reduce the frequency of finger-stick testing (the Dexcom G5 system, for example, requires two daily sticks). To some degree, however, the devices remain invasive.
Viable Alternatives
Today, after years of false starts, the prospect of pain-free glucose monitoring may finally be at hand. With a whole host of non-invasive glucose monitoring (NIGM) devices now under development—many on the verge of market entry—diabetics will be eagerly scrutinizing their pros and cons.
NIGM devices monitor glucose levels without compromising the skin barrier. They either provide continuous readings similar to CGM devices, or intermittent readings where the wearer has to perform some kind of manual test.
A recent report by the University of Birmingham identified 40 new and emerging NIGM technologies, using optical, transdermal or electrochemical techniques. Potential sites for glucose testing included skin, tear fluid, saliva and breath.
Beat This
Following regulatory (CE) approval, U.K.-based Nemaura Medical plans to introduce its SugarBEAT continuous NIGM system to markets in Europe, Asia and the Middle East in 2017. A self-applied system, it relies on a daily disposable patch to draw interstitial fluid to the surface of the skin via weak electric currrents. Fluid in the patch “reservoir” is then analyzed for glucose by a reusable, electronic sensor.
With SugarBEAT’s sensor activating every five minutes, data is sent to a smartphone, smartwatch or stand-alone reader by Bluetooth for processing. From here it can easily be forwarded to medical professionals. The system requires one daily finger-stick test for calibration, and provides audible alerts.
Due to the lag time between changes in plasma and interstitial glucose levels, CGM systems (both invasive and non-invasive) are less accurate than the finger stick technique. Invasive measurement systems typically use MARD (mean absolute relative deviation) as a measure of accuracy.
Dexcom’s G5, which has a MARD of less than 10%, is generally considered the benchmark for CGM accuracy. By way of comparison, SugarBEAT’s MARD ranged from 14% to 18% in recent trials.
One of our primary goals has always been affordability
“This is deemed adequate for its intended use, since a confirmatory finger prick is still required when therapy decisions need to be made,” says Nemaura Medical CEO Dr. Faz Chowdhury. “Going forward, improved SugarBEAT sensor performance should see MARD continue to fall.”
While no price point has yet been announced, Chowdhury claims SugarBEAT will be the cheapest CGM on the market, with disposable patches costing a few dollars each.
“One of our primary goals has always been affordability,” he says.
Aural Approach
Going one step further on the finger prick reduction front is GlucoTrack. A non-continuous NIGM system from Israeli firm Integrity Applications, it only requires six finger stick tests per year.
With widespread market entry in 2016, GlucoTrack has already gained regulatory approval in Europe and South Korea, and is working toward approval in the U.S. and China. The company claims accuracy is “equivalent or better than that of other commercially available CGM systems.”
GlucoTrack uses an earlobe clip-on sensor similar to a pulse oximeter to analyze glucose levels through the skin, using ultrasound, electromagnetic and thermal technologies. The abundance of capillaries and relatively slow blood flow in the earlobe apparently helps to give more accurate readings than at other body testing sites.
GlucoTrack is the only truly non-invasive device available in the world
“GlucoTrack is the only truly non-invasive device available in the world,” claims Integrity CEO Avner Gal. “It is also the only device that combines multiple sensors and technologies to increase reliability.”
Readings from GlucoTrack’s clip-on sensor are transferred via cable to a smartphone-sized “module” that calculates glucose levels. Processed data can then be downloaded for professional analysis (wireless functionality for the module is now under development). While the sensor has to be calibrated every six months with three finger pricks (carried out at the point of sale or in a clinic), it can be used as often as required, and readings take less than a minute.
First-year costs for the latest GlucoTrack model are around $2,100, including one replaceable sensor. This compares favorably with current prices for Dexcom’s G5 (including transmitters and sensors).
With the demand for affordable, practical, accurate and painless glucose monitoring devices at an all-time high, SugarBEAT and GlucoTrack are just two of the many NIGM devices currently making waves. If these devices can be shown to improve glycemic control, then finger prick testing may one day become a pain of the past.
Learn more on continuous blood glucose meters on MedicalExpo.
https://www.youtube.com/watch?v=4ezjIxqFIxQ















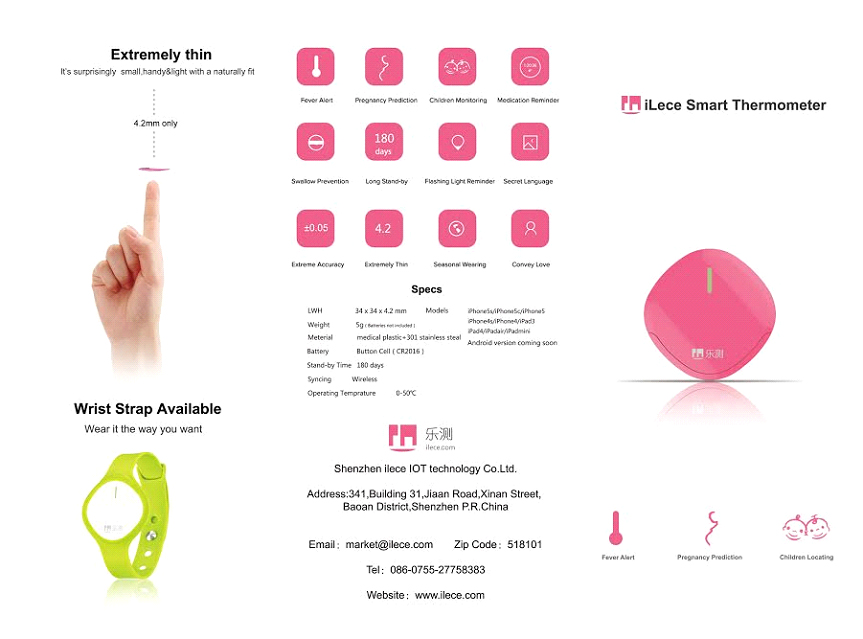
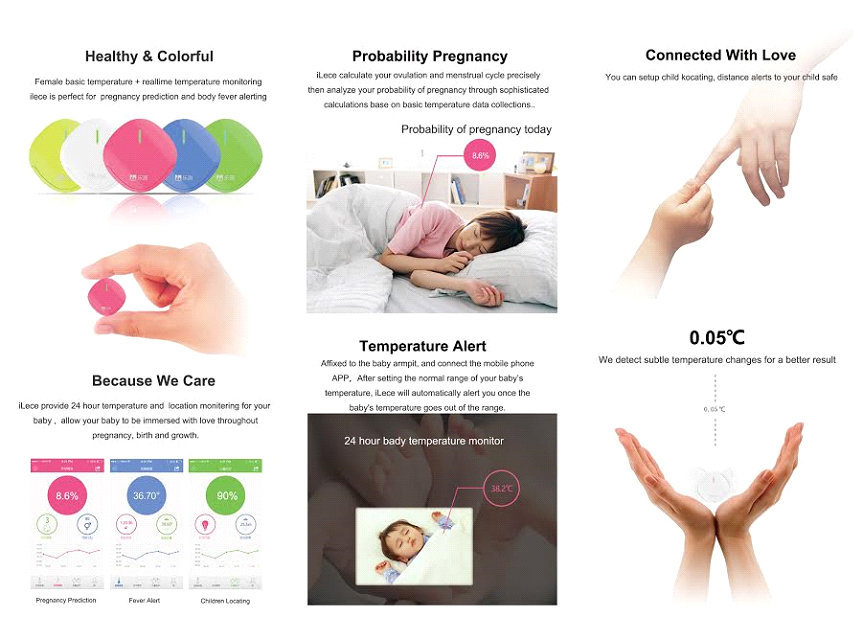
 Cardiovascular diseases, respiratory diseases and sleeping problems are very common and can occur at any age. Early detection of symptoms should mean early medical consultation and early medical diagnosis for effective prevention or lifesaving early intervention.
Cardiovascular diseases, respiratory diseases and sleeping problems are very common and can occur at any age. Early detection of symptoms should mean early medical consultation and early medical diagnosis for effective prevention or lifesaving early intervention.



 A social program thought and designed to help people.
This app can be used for simple investigations (check temperature, monitoring the evolution of diabetes or blood pressure)
A social program thought and designed to help people.
This app can be used for simple investigations (check temperature, monitoring the evolution of diabetes or blood pressure) 
